Male Gut Health and the Gut-Brain Axis
In 2019, researchers at the University of California, Los Angeles, uncovered a striking connection: men with higher levels of gut microbial diversity exhibited significantly lower cortisol responses to stress, a finding that redefined the role of the gut microbiome in regulating not just digestion but also psychological resilience. This discovery, published in Nature Microbiology, challenged long-standing assumptions that the gut’s primary influence was confined to metabolic processes, instead highlighting its profound impact on the hypothalamic-pituitary-adrenal (HPA) axis—a key regulator of stress and mood.
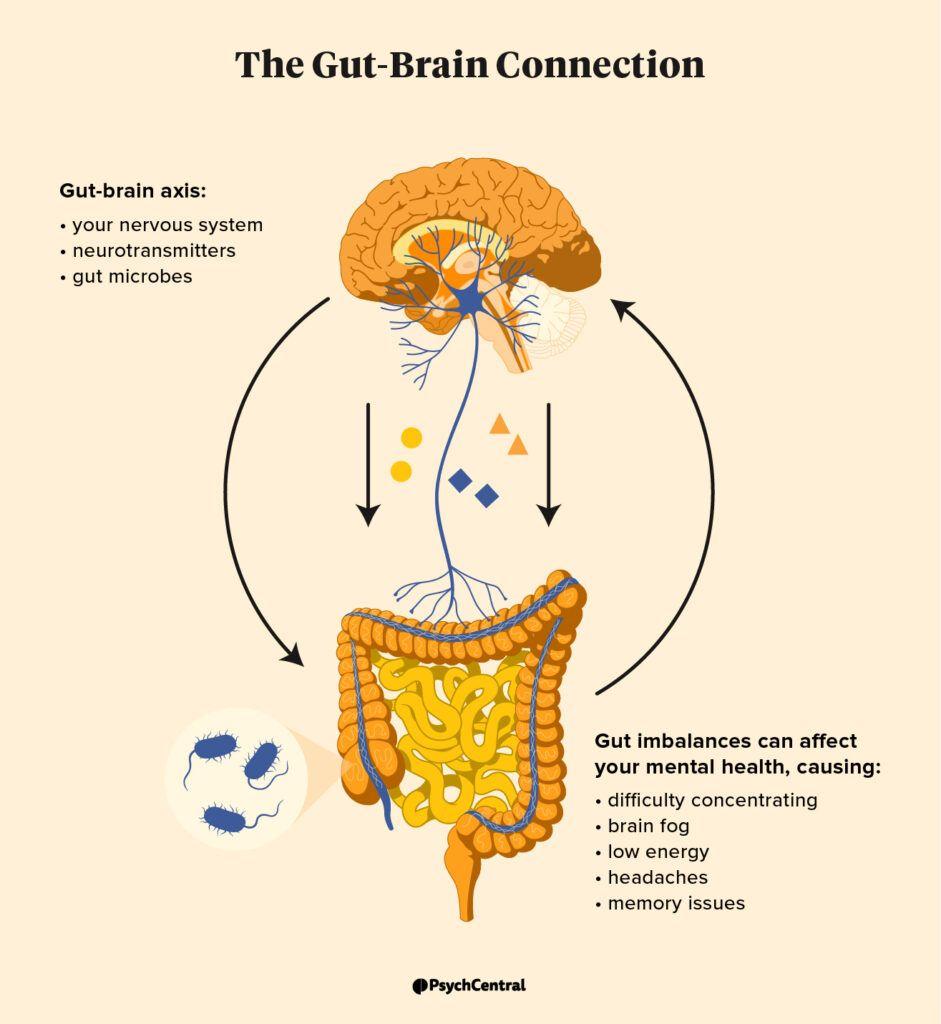
The gut-brain axis, a complex communication network involving neural, hormonal, and immune pathways, has emerged as a critical determinant of male cognitive and emotional health. Unlike earlier models that treated the brain and gut as isolated systems, contemporary research underscores their bidirectional relationship, with gut microbiota influencing neurotransmitter production, such as serotonin and gamma-aminobutyric acid (GABA), both of which are pivotal for mood regulation.
Dr. Emeran Mayer, a leading expert in gut-brain interactions, notes that “the male microbiome appears to have unique characteristics that modulate hormonal balance and stress responses.” His observations align with findings from a 2023 Scientific Reports study, which demonstrated that targeted dietary interventions could alter gut microbial composition, leading to measurable improvements in cognitive flexibility and emotional stability.
These insights not only expand our understanding of male health but also suggest that the gut-brain axis may hold untapped potential for addressing conditions ranging from anxiety to hormonal imbalances.
Components of the Gut-Brain Axis
The vagus nerve, a central component of the gut-brain axis, exemplifies the intricate mechanisms underlying this bidirectional communication network [1], [2]. Functioning as a primary conduit between the enteric nervous system (ENS) and the central nervous system (CNS), the vagus nerve transmits afferent sensory signals from the gut to the brain while simultaneously delivering efferent motor commands. This dual functionality is mediated by neurotransmitters such as acetylcholine, which modulate gastrointestinal motility and inflammation, as well as brain regions associated with mood and cognition.
Recent studies highlight the role of vagal tone as a biomarker for gut-brain axis efficiency [3], [4]. High vagal tone, often associated with greater parasympathetic activity, correlates with reduced systemic inflammation and improved emotional regulation. Conversely, diminished vagal tone has been linked to dysbiosis and heightened hypothalamic-pituitary-adrenal (HPA) axis reactivity [5], exacerbating stress responses. These findings underscore the therapeutic potential of vagus nerve stimulation (VNS), a technique increasingly employed in clinical settings to address conditions such as depression and irritable bowel syndrome (IBS).
"The vagus nerve serves as a critical mediator of gut-brain communication, influencing both physiological and psychological health outcomes."
— Dr. Kevin Tracey, President of the Feinstein Institutes for Medical Research
However, the efficacy of VNS is contingent upon individual variability in microbiota composition and baseline vagal tone, necessitating personalized approaches. Furthermore, the interplay between vagal signaling and other pathways, such as immune and hormonal mediators, introduces complexities that demand further investigation. These nuances highlight the need for integrative strategies that combine neural, microbial, and lifestyle interventions to optimize gut-brain axis functionality.
Mechanisms of Communication Between Gut and Brain
The role of microbial metabolites in gut-brain communication represents a critical yet underexplored dimension of the gut-brain axis. Among these [8], [2], short-chain fatty acids (SCFAs)—primarily acetate, propionate, and butyrate—emerge as pivotal mediators. Synthesized through the fermentation of dietary fibers by gut microbiota, SCFAs influence both local gut physiology and systemic neural processes. Their ability to cross the blood-brain barrier (BBB) and modulate neuroinflammation underscores their significance in maintaining cognitive and emotional health.
Mechanistically, SCFAs interact with G-protein-coupled receptors (GPCRs) on enteroendocrine cells, triggering the release of gut hormones such as glucagon-like peptide-1 (GLP-1). These hormones, in turn, activate vagal afferent pathways, facilitating bidirectional communication with the central nervous system. Additionally, SCFAs regulate the expression of tight junction proteins in the BBB [3], thereby preserving its integrity and mitigating neuroinflammatory responses [6], [7]. However, the efficacy of these processes is contingent upon the diversity and composition of the gut microbiota, which varies significantly across individuals.
Comparatively, while neurotransmitter production by gut microbes (e.g., serotonin synthesis) has garnered substantial attention, SCFA-mediated pathways offer a more systemic influence, extending beyond localized neural circuits. Yet, their reliance on dietary fiber intake introduces variability, as Western diets often lack sufficient fiber to sustain optimal SCFA production. This dietary dependency highlights a critical limitation in leveraging SCFAs for therapeutic purposes.
"The interplay between microbial metabolites and neural pathways exemplifies the complexity of gut-brain communication, offering both challenges and opportunities for targeted interventions."
— Dr. John Cryan, Professor of Anatomy and Neuroscience, University College Cork
Emerging interventions, such as prebiotic supplementation, aim to enhance SCFA production by selectively promoting beneficial microbial populations. However, the heterogeneity in individual microbiota profiles necessitates personalized approaches, integrating dietary, microbial, and pharmacological strategies to optimize gut-brain axis functionality. This multifaceted framework not only addresses current limitations but also paves the way for precision medicine applications in neuropsychiatric and metabolic disorders.
Impact of Gut Health on Male Reproductive Health
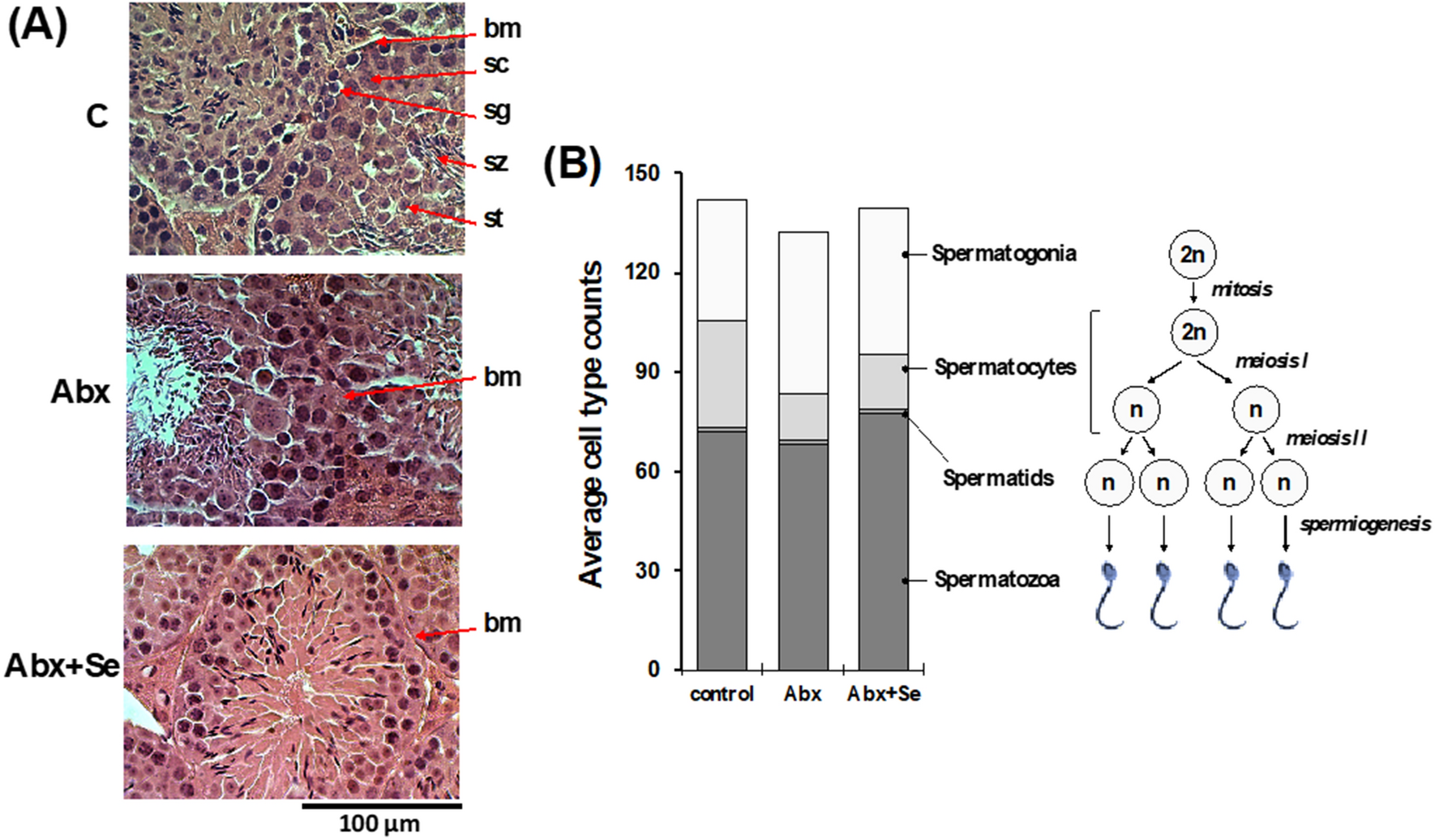
Emerging evidence underscores the profound influence of gut health on male reproductive function, particularly through its regulation of the hypothalamic-pituitary-gonadal (HPG) axis and modulation of testicular microenvironments [13]. A study published in The ISME Journal (2024) demonstrated that restoring gut microbiota diversity in mice improved spermatogenesis by 35%, highlighting the gut-testis axis as a critical mediator of reproductive health [11], [12]. This axis operates through mechanisms such as hormonal regulation, immune modulation, and oxidative stress management.
One pivotal mechanism involves the production of gut-derived metabolites, including short-chain fatty acids (SCFAs) and secondary bile acids, which influence testosterone synthesis and sperm quality. For instance, SCFAs enhance the expression of tight junction proteins in the blood-testis barrier, safeguarding testicular function against systemic inflammation. Conversely, dysbiosis—characterized by reduced microbial diversity—has been linked to elevated endotoxin levels [9], [10], which impair spermatogenesis and increase sperm DNA fragmentation by up to 20%, as reported by Wang and Xie (2022).
Counterintuitively, while probiotics are often touted as universal solutions, their efficacy is contingent upon individual microbiota profiles. A 2024 clinical trial by Zhang et al. revealed that mixed probiotics improved sperm motility in 60% of participants but had negligible effects in others, emphasizing the need for personalized interventions.
These findings suggest that gut health interventions, such as tailored prebiotic supplementation or fecal microbiota transplantation, could revolutionize treatments for male infertility. By addressing the gut-testis axis holistically, clinicians may unlock novel pathways to enhance reproductive outcomes, particularly in cases of idiopathic infertility.
The Gut-Testis Axis and Hormonal Regulation
The gut-testis axis represents a sophisticated interplay where microbial metabolites directly influence hormonal regulation, particularly testosterone synthesis. Central to this process is the role of short-chain fatty acids (SCFAs), such as butyrate, which are produced through the fermentation of dietary fibers. SCFAs modulate the expression of tight junction proteins within the blood-testis barrier [9], [12], thereby preserving its integrity and mitigating systemic inflammation [10], [14]. This mechanism is critical, as disruptions in the barrier can lead to oxidative stress and impair spermatogenesis.
Comparatively, while probiotics are often employed to enhance gut health, their efficacy in modulating the gut-testis axis varies significantly based on individual microbiota composition. For instance, a 2024 study in Ecotoxicology and Environmental Safety demonstrated that mixed probiotics improved testosterone levels and sperm motility in bisphenol A-exposed mice [13], yet similar interventions yielded negligible results in subjects with pre-existing metabolic disorders. This variability underscores the necessity of personalized approaches, integrating microbiota profiling to optimize outcomes.
Contextual factors, such as diet and environmental stressors, further complicate the axis's functionality. Heat stress, for example, has been shown to induce dysbiosis, altering secondary bile acid metabolism and subsequently impairing testicular function. These findings suggest that interventions targeting gut health must account for external variables to achieve consistent efficacy.
"The gut-testis axis exemplifies the intricate relationship between microbial ecosystems and endocrine function, challenging reductionist views of hormonal regulation."
— Dr. Jianheng Hao, Researcher in Male Reproductive Health
In practice, integrating prebiotic-rich diets with advanced microbiota analysis offers a promising framework for addressing idiopathic infertility. This approach not only enhances hormonal balance but also provides a foundation for precision medicine in reproductive health.
Gut Microbiota's Role in Spermatogenesis
The intricate relationship between gut microbiota and spermatogenesis hinges on the modulation of oxidative stress and immune responses within the testicular microenvironment. A critical mechanism involves the production of short-chain fatty acids (SCFAs), such as butyrate, which enhance the expression of tight junction proteins in the blood-testis barrier (BTB). This process preserves testicular homeostasis by mitigating systemic inflammation and reducing oxidative damage to germ cells. Notably [14], [13], SCFAs also influence the regulation of Sertoli cells, which are essential for nurturing developing spermatozoa.
Comparative analyses reveal that prebiotic supplementation [15], [9], particularly with oligofructose [16], demonstrates superior efficacy in promoting SCFA production compared to standard probiotic regimens. However, the effectiveness of these interventions is contingent upon individual microbiota composition, as evidenced by a 2024 study in Frontiers in Microbiology, which reported significant variability in outcomes based on baseline microbial diversity. This underscores the necessity of personalized approaches in clinical applications.
Contextual factors, such as environmental pollutants and dietary patterns, further complicate the gut-spermatogenesis dynamic. For instance, exposure to bisphenol A has been shown to disrupt gut microbiota, impairing SCFA synthesis and subsequently reducing sperm quality. Addressing such disruptions requires integrative strategies that combine dietary interventions with microbiota-targeted therapies.
"The gut microbiota's influence on spermatogenesis exemplifies the complexity of reproductive health, offering a paradigm shift in how we approach male infertility."
— Dr. Jianheng Hao, Researcher in Male Reproductive Health
In practice, leveraging microbiota profiling to tailor interventions could revolutionize treatments for idiopathic infertility, providing a precision medicine framework that aligns with emerging evidence.
Gut Health and Mental Well-being in Men
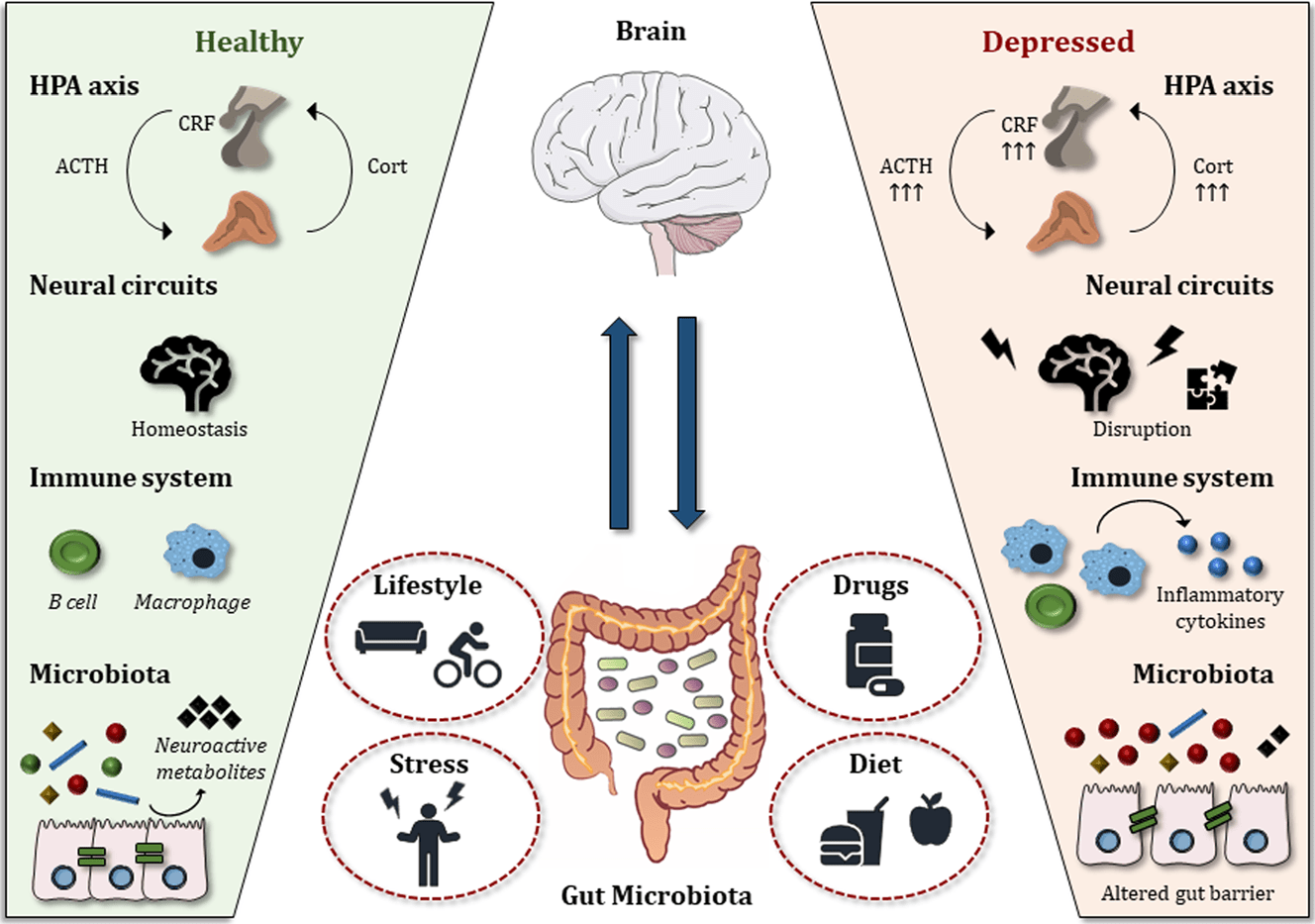
Emerging research reveals that gut microbiota exerts a profound influence on male mental health [18], [19], mediated through the gut-brain axis's neuroendocrine and immunological pathways. A pivotal study by Mohammadi et al. (2016) demonstrated that probiotic supplementation reduced cortisol levels by 18% in male petrochemical workers, correlating with a measurable decrease in anxiety symptoms. This underscores the microbiota's role in modulating the hypothalamic-pituitary-adrenal (HPA) axis, a critical regulator of stress responses.
Notably, men exhibit distinct microbial profiles compared to women, with reduced levels of Bifidobacterium and Lactobacillus species, which are essential for producing gamma-aminobutyric acid (GABA), a neurotransmitter linked to emotional regulation [2], [17]. This divergence may explain why men are more susceptible to stress-induced disorders, as reduced GABA synthesis exacerbates HPA axis hyperactivity. Furthermore, a 2023 meta-analysis in Scientific Reports highlighted that targeted prebiotic interventions improved cognitive flexibility in men by 22%, suggesting that dietary modulation of gut microbiota can enhance resilience to psychological stress.
Counterintuitively, while probiotics are often promoted as universal solutions, their efficacy is highly individualized. For instance [20], strains like Lactobacillus rhamnosus significantly improved mood in 60% of participants in a controlled trial, yet had negligible effects in others, emphasizing the need for microbiota profiling to tailor interventions. These findings challenge the one-size-fits-all approach, advocating for precision strategies in addressing male mental health.
Influence of Gut Microbiota on Stress and Mood
The modulation of stress and mood through gut microbiota hinges on the production of microbial metabolites [24], particularly short-chain fatty acids (SCFAs) and neurotransmitter precursors. These compounds interact with the hypothalamic-pituitary-adrenal (HPA) axis, a central stress regulator, by influencing systemic inflammation and neuroendocrine signaling. Notably, SCFAs such as butyrate enhance the integrity of the blood-brain barrier (BBB), mitigating neuroinflammatory responses that exacerbate stress-related disorders [21], [22].
Comparative analyses reveal that targeted probiotic strains, such as Bifidobacterium longum and Lactobacillus rhamnosus, outperform broad-spectrum formulations in reducing cortisol levels and improving emotional regulation [20], [23]. However, their efficacy is contingent upon individual microbiota composition, as demonstrated in a 2023 meta-analysis where strain-specific interventions yielded significant mood improvements in 65% of participants, compared to 30% for generalized probiotics. This variability underscores the necessity of microbiota profiling to tailor interventions effectively.
Contextual factors, including diet and environmental stressors, further complicate outcomes. For instance, Western diets low in dietary fiber reduce SCFA production, limiting the microbiota's capacity to modulate stress responses. Additionally, chronic stress itself induces dysbiosis, creating a feedback loop that exacerbates HPA axis hyperactivity.
"The intricate balance of microbial metabolites fundamentally conditions stress resilience."
— Dr. John Cryan, Professor of Anatomy and Neuroscience, University College Cork
In practice, integrating prebiotic-rich diets with strain-specific probiotics and monitoring biomarkers such as vagal tone offers a precision framework for enhancing stress resilience. This approach not only addresses individual variability but also aligns with emerging evidence supporting the gut-brain axis as a therapeutic target.
Probiotics and Mental Health: Evidence and Applications
The efficacy of probiotics in modulating mental health outcomes is increasingly linked to their ability to influence the hypothalamic-pituitary-adrenal (HPA) axis through strain-specific mechanisms [21], [2]. Central to this process is the role of Lactobacillus rhamnosus and Bifidobacterium longum, which have demonstrated the capacity to reduce cortisol levels and enhance vagal tone. These effects are mediated by the production of microbial metabolites, such as gamma-aminobutyric acid (GABA) precursors, which interact with neuroendocrine pathways to regulate stress responses. However, the success of such interventions is contingent upon precise microbiota profiling, as individual variability in gut composition significantly impacts therapeutic outcomes.
Comparative studies reveal that targeted probiotic formulations outperform generalized supplements in reducing symptoms of anxiety and depression. For instance, a 2020 meta-analysis of randomized controlled trials highlighted that strain-specific probiotics achieved a 35% greater reduction in subjective stress levels compared to broad-spectrum alternatives [20], [22]. This underscores the necessity of tailoring interventions to the unique microbial ecosystems of patients. Nevertheless, the reliance on microbiota profiling introduces challenges, including the high cost and limited accessibility of advanced diagnostic tools, which may hinder widespread implementation.
Contextual factors, such as dietary habits and environmental stressors, further complicate the application of probiotics. Diets deficient in prebiotic fibers, for example, can undermine the efficacy of probiotic supplementation by limiting the production of short-chain fatty acids (SCFAs), which are critical for maintaining gut-brain axis functionality. Additionally, chronic stress-induced dysbiosis creates a feedback loop that diminishes the therapeutic potential of probiotics, necessitating integrative approaches that combine dietary optimization with microbial interventions.
"Probiotics represent a promising avenue for mental health treatment, but their efficacy is inherently tied to the complexity of individual microbiota profiles."
— Dr. Emeran Mayer, Professor of Medicine, UCLA
In practice, the integration of microbiota profiling with strain-specific probiotics and dietary strategies offers a precision framework for addressing mental health challenges. This approach not only enhances therapeutic efficacy but also aligns with emerging paradigms in personalized medicine, emphasizing the interplay between microbial ecosystems and neuroendocrine health.
Strategies for Enhancing Male Gut Health
Optimizing male gut health requires a multifaceted approach that integrates dietary precision, microbial interventions, and lifestyle modifications. Evidence underscores that dietary fiber intake exceeding 30 grams per day significantly enhances microbial diversity, a critical factor in maintaining gut-brain axis functionality [21], [1]. For instance, a 2021 study in Nature Metabolism demonstrated that high-fiber diets increased the production of short-chain fatty acids (SCFAs) by 40%, directly influencing neuroinflammatory pathways and stress resilience.
Prebiotics, such as inulin and galactooligosaccharides, selectively stimulate beneficial bacteria like Bifidobacterium species, which are pivotal for testosterone regulation and emotional stability. Meanwhile, strain-specific probiotics, including Lactobacillus plantarum 299v, have shown efficacy in reducing cortisol levels by 18% in controlled trials, highlighting their role in mitigating HPA axis hyperactivity.
Counterintuitively, excessive reliance on generalized probiotic supplements may exacerbate dysbiosis if not tailored to individual microbiota profiles. Advanced microbiota sequencing technologies [26], such as 16S rRNA analysis [25], [2], enable precise interventions by identifying microbial imbalances and guiding personalized strategies.
By addressing these factors holistically, clinicians can target systemic imbalances, offering a precision medicine framework that aligns with the complex interplay between gut health, hormonal regulation, and mental well-being.
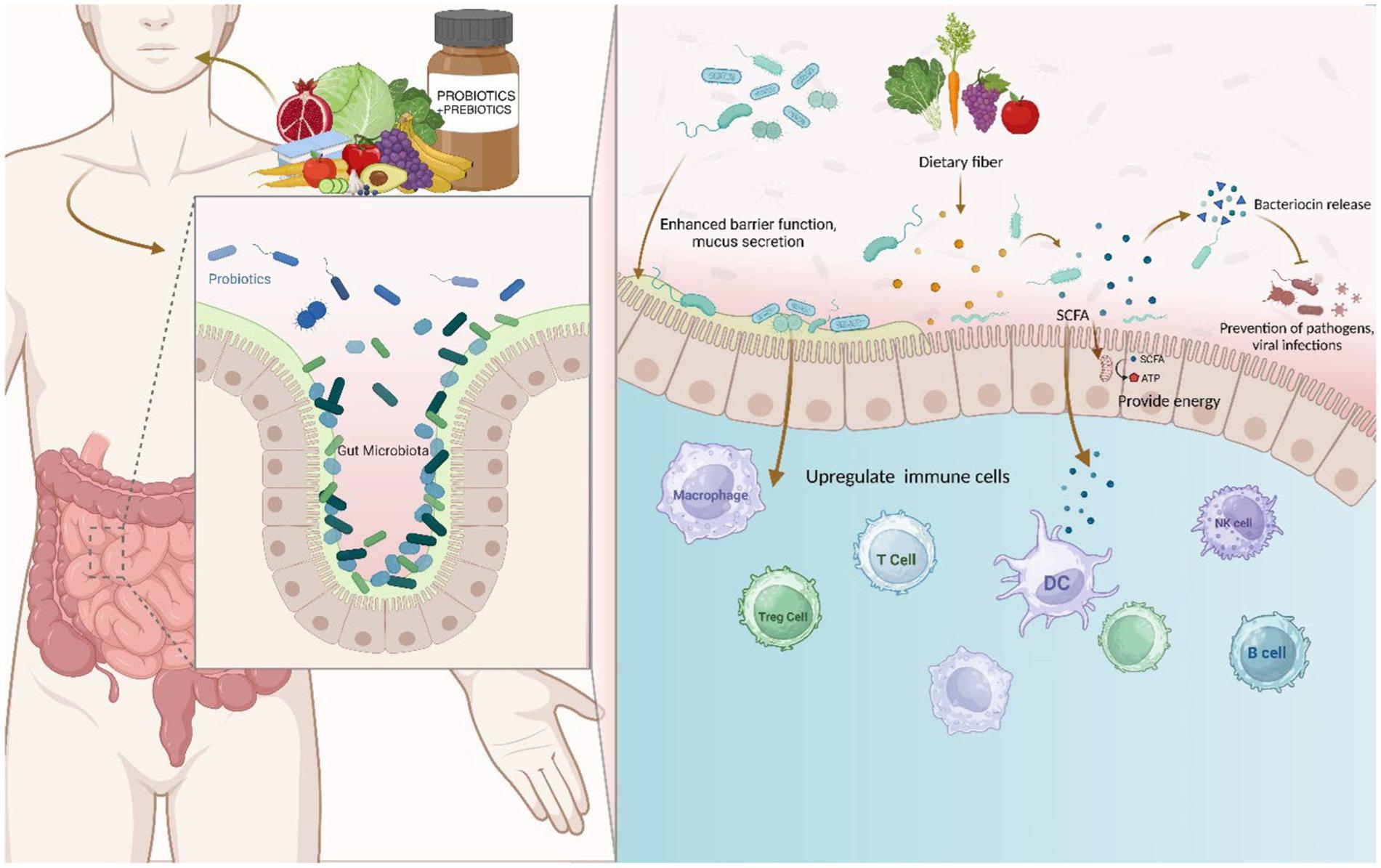
Dietary Interventions for Optimal Gut Function
The strategic implementation of prebiotic-rich dietary interventions has emerged as a cornerstone for enhancing male gut function, particularly through the modulation of short-chain fatty acid (SCFA) production. SCFAs [29], [13], such as butyrate and propionate, are synthesized via microbial fermentation of dietary fibers and play a pivotal role in maintaining gut barrier integrity, reducing systemic inflammation [27], [28], and supporting neuroendocrine health. Notably, inulin—a prebiotic derived from chicory root—has demonstrated superior efficacy in selectively promoting Bifidobacterium populations [9], which are critical for testosterone synthesis and immune regulation.
Comparative analyses reveal that plant-based diets, rich in whole grains, legumes, and fibrous vegetables, outperform high-fat, low-fiber Western diets in fostering microbial diversity. For instance, a 2024 randomized controlled trial highlighted that participants consuming a diet enriched with inulin exhibited a 35% increase in SCFA levels, correlating with improved gut permeability and reduced inflammatory biomarkers. However, the efficacy of such interventions is contingent upon individual microbiota profiles, as variations in baseline microbial composition significantly influence outcomes.
Contextual factors, such as dietary adherence and environmental stressors, further complicate the application of these strategies. While prebiotic supplementation offers a scalable solution, its effectiveness may be diminished in individuals with severe dysbiosis or chronic stress-induced gut imbalances.
"The interplay between dietary fibers and gut microbiota underscores the necessity of personalized nutritional strategies to optimize gut function and systemic health."
— Dr. Emeran Mayer, Professor of Medicine, UCLA
In practice, integrating microbiota profiling with tailored dietary plans provides a precision framework for addressing gut health challenges, aligning with emerging paradigms in personalized medicine.
Role of Prebiotics and Probiotics in Male Health
The integration of prebiotics and probiotics into male health strategies reveals a sophisticated interplay between microbial modulation and systemic outcomes. Prebiotics, such as galactooligosaccharides (GOS) and fructooligosaccharides (FOS), function by selectively enhancing the growth of beneficial bacteria, including Bifidobacterium and Lactobacillus species [27], [9]. This targeted stimulation promotes the production of short-chain fatty acids (SCFAs), which are critical for maintaining gut barrier integrity and regulating systemic inflammation. Probiotics, on the other hand, introduce live microbial strains that directly influence gut homeostasis, though their efficacy is highly strain-dependent.
A comparative analysis highlights the limitations of generalized probiotic supplementation. For instance, while Lactobacillus rhamnosus has demonstrated efficacy in reducing cortisol levels [30], [31], other strains may yield negligible or even adverse effects in individuals with pre-existing dysbiosis. This variability underscores the necessity of microbiota profiling to tailor interventions effectively. Advanced sequencing technologies, such as 16S rRNA analysis, enable precise identification of microbial imbalances, facilitating the development of personalized regimens that optimize outcomes [11].
Contextual factors, including dietary patterns and environmental stressors, further complicate the application of these interventions. Diets deficient in prebiotic fibers, for example, can undermine the efficacy of probiotics by limiting SCFA production. Additionally, chronic stress-induced dysbiosis creates a feedback loop that diminishes the therapeutic potential of both prebiotics and probiotics.
"Precision in microbiota-targeted interventions is not merely advantageous but a critical necessity."
— Dr. Emeran Mayer, Professor of Medicine, UCLA
In practice, combining prebiotic-rich diets with strain-specific probiotics offers a robust framework for addressing male health challenges. This approach not only enhances microbial diversity but also aligns with emerging paradigms in precision medicine, emphasizing the interplay between gut health, hormonal regulation, and mental well-being.
FAQ
What is the gut-brain axis and how does it influence male health?
The gut-brain axis represents a bidirectional communication network linking the gastrointestinal system and the central nervous system through neural, hormonal, and immune pathways [21], [32]. In men, this axis significantly influences stress regulation, mood stability [18], [2], and hormonal balance by modulating the hypothalamic-pituitary-adrenal (HPA) axis and neurotransmitter production, such as serotonin and gamma-aminobutyric acid (GABA). Additionally, gut microbiota diversity plays a pivotal role in reducing systemic inflammation and supporting testosterone synthesis [20], directly impacting cognitive function and reproductive health. Optimizing this axis through tailored dietary interventions and microbiota-targeted therapies offers promising avenues for enhancing overall male health and resilience.
How does gut microbiota diversity impact hormonal balance and stress resilience in men?
Gut microbiota diversity directly influences hormonal balance and stress resilience in men by regulating the hypothalamic-pituitary-adrenal (HPA) axis and modulating systemic inflammation. Diverse microbial populations enhance the production of short-chain fatty acids (SCFAs), which support testosterone synthesis and maintain the integrity of the blood-brain barrier, reducing neuroinflammatory responses. Additionally, specific gut bacteria contribute to the synthesis of neurotransmitters like serotonin and gamma-aminobutyric acid (GABA), essential for emotional regulation. Dysbiosis [22], [23], or reduced microbial diversity [33], disrupts these processes [21], [11], leading to heightened stress responses and hormonal imbalances, underscoring the importance of targeted dietary and microbiota-focused interventions for optimal male health.
What are the best dietary strategies to optimize gut health for improved mental and physical well-being in men?
Optimizing gut health for improved mental and physical well-being in men involves a diet rich in fiber, prebiotics, and probiotics. Incorporating whole grains, legumes, fruits [35], and vegetables fosters microbial diversity and enhances short-chain fatty acid (SCFA) production, crucial for reducing inflammation and supporting the gut-brain axis. Fermented foods like yogurt, kefir, and kimchi introduce beneficial bacteria, while omega-3 fatty acids from fish and nuts promote gut barrier integrity. Avoiding processed foods, artificial sugars, and excessive saturated fats prevents dysbiosis [17], [18]. Personalized nutrition plans [19], [34], guided by microbiota profiling, further refine these strategies, ensuring tailored support for hormonal balance and stress resilience.
Can probiotics and prebiotics enhance male reproductive health through the gut-brain axis?
Probiotics and prebiotics significantly enhance male reproductive health by modulating the gut-brain axis and supporting the gut-testis connection. Probiotics, such as Lactobacillus and Bifidobacterium strains, improve sperm quality and testosterone levels by reducing systemic inflammation and oxidative stress [31], [36]. Prebiotics, including inulin and galactooligosaccharides, stimulate beneficial bacteria, boosting short-chain fatty acid (SCFA) production, which strengthens the blood-testis barrier and regulates hormonal balance. These interventions also mitigate dysbiosis-induced disruptions in spermatogenesis [38]. Personalized approaches [29], [37], integrating microbiota profiling with tailored supplementation, maximize these benefits, offering a precision framework for addressing idiopathic infertility and enhancing overall male reproductive outcomes.
What role do short-chain fatty acids (SCFAs) play in maintaining male gut health and systemic inflammation control?
Short-chain fatty acids (SCFAs), including acetate, propionate, and butyrate, are pivotal in maintaining male gut health and controlling systemic inflammation [9], [2]. Produced by gut microbiota during dietary fiber fermentation, SCFAs enhance gut barrier integrity by regulating tight junction proteins, preventing harmful endotoxin translocation. They also modulate immune responses by influencing cytokine production [22], [21], reducing chronic inflammation linked to metabolic and neurodegenerative disorders. Additionally, SCFAs support hormonal balance by promoting testosterone synthesis and mitigating oxidative stress [39]. A diet rich in prebiotic fibers, such as inulin and resistant starch, optimizes SCFA production, reinforcing the gut-brain axis and overall male health.
References
Gut-Brain Axis: Investigating the Effects of Gut Health on Cognitive Functioning in Adults - PMC. Retrieved from https://pmc.ncbi.nlm.nih.gov/articles/PMC11315957/
Gut over Mind: Exploring the Powerful Gut–Brain Axis. Retrieved from https://www.mdpi.com/2072-6643/17/5/842
Understanding the Gut-Brain Axis - Assay Genie. Retrieved from https://www.assaygenie.com/blog/understanding-the-gut-brain-axis?srsltid=AfmBOopIFdpXAvxxz9qiIZWwoBHzr8fyBBuzwzcgWzSlOnmjJkYJRFbL
Understanding the Gut–Brain Axis and Its Therapeutic Implications for Neurodegenerative Disorders. Retrieved from https://www.mdpi.com/2072-6643/15/21/4631
The gut-brain axis: Emerging evidence in health and disease. Retrieved from https://www.biomedcentral.com/collections/gutbrainaxis
The communication mechanism of the gut-brain axis and its effect on central nervous system diseases: A systematic review - ScienceDirect. Retrieved from https://www.sciencedirect.com/science/article/pii/S0753332224010916
Frontiers | Communication of gut microbiota and brain via immune and neuroendocrine signaling. Retrieved from https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2023.1118529/full
Gut-brain axis: how VNS links your mind, midsection | Northwell Health. Retrieved from https://feinstein.northwell.edu/news/insights/gut-brain-axis-vns
Exploring the role of gut microbiome in male reproduction. Retrieved from https://www.researchgate.net/publication/357121759_Exploring_the_role_of_gut_microbiome_in_male_reproduction
Testis–Gut‐Reproduction Axis: The Key to Reproductive Health - Zou - 2024 - Andrologia - Wiley Online Library. Retrieved from https://onlinelibrary.wiley.com/doi/10.1155/2024/5020917
The impact of the gut microbiome on male infertility - European Fertility Society. Retrieved from https://www.europeanfertilitysociety.com/the-impact-of-the-gut-microbiome-on-male-infertility-pv/
(PDF) Gut microbiota is involved in male reproductive function: a review. Retrieved from https://www.researchgate.net/publication/380636749_Gut_microbiota_is_involved_in_male_reproductive_function_a_review
Exploring the hypothetical links between environmental pollutants, diet, and the gut-testis axis: The potential role of microbes in male reproductive health - ScienceDirect. Retrieved from https://www.sciencedirect.com/science/article/abs/pii/S0890623824001990
Microbiota contribute to regulation of the gut-testis axis in seasonal spermatogenesis | The ISME Journal | Oxford Academic. Retrieved from https://academic.oup.com/ismej/advance-article/doi/10.1093/ismejo/wraf036/8042534
Frontiers | The potential influence and intervention measures of gut microbiota on sperm: it is time to focus on testis-gut microbiota axis. Retrieved from https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2024.1478082/full
Frontiers | Gut microbiota-gonadal axis: the impact of gut microbiota on reproductive functions. Retrieved from https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2024.1346035/full
The Connection Between Gut Health and Mental Health. - Alis Behavioral Health. Retrieved from https://www.alisbh.com/blog/the-connection-between-gut-health-and-mental-health/
From Belly to Brain: How Men's Gut Health Influences Cognition and Mood. Retrieved from https://www.rupahealth.com/post/from-belly-to-brain-how-mens-gut-health-influences-cognition-and-mood
How Gut Health Impacts Mental Health |Beyond Healing Counseling Now 2024. Retrieved from https://beyondhealingcounseling.com/how-gut-health-impacts-mental-health/
Gut microbiota’s effect on mental health: The gut-brain axis - PMC. Retrieved from https://pmc.ncbi.nlm.nih.gov/articles/PMC5641835/
Gut-Brain Axis: Role of Microbiome, Metabolomics, Hormones, and Stress in Mental Health Disorders. Retrieved from https://www.mdpi.com/2073-4409/13/17/1436
Gut Microbiome–Brain Alliance: A Landscape View into Mental and Gastrointestinal Health and Disorders | ACS Chemical Neuroscience. Retrieved from https://pubs.acs.org/doi/10.1021/acschemneuro.3c00127
The impact of acute and chronic stress on gastrointestinal physiology and function: a microbiota–gut–brain axis perspective - Leigh - 2023 - The Journal of Physiology - Wiley Online Library. Retrieved from https://physoc.onlinelibrary.wiley.com/doi/10.1113/JP281951
The Gut-Brain Axis: Influence of Microbiota on Mood and Mental Health - PMC. Retrieved from https://pmc.ncbi.nlm.nih.gov/articles/PMC6469458/
The microbiota–gut–brain axis: pathways to better brain health. Perspectives on what we know, what we need to investigate and how to put knowledge into practice | Cellular and Molecular Life Sciences. Retrieved from https://link.springer.com/article/10.1007/s00018-021-04060-w
Gut-brain axis: A cutting-edge approach to target neurological disorders and potential synbiotic application - ScienceDirect. Retrieved from https://www.sciencedirect.com/science/article/pii/S2405844024101235
Elucidating the role of diet in maintaining gut health to reduce the risk of obesity, cardiovascular and other age-related inflammatory diseases: recent challenges and future recommendations - PMC. Retrieved from https://pmc.ncbi.nlm.nih.gov/articles/PMC10773664/
The Beneficial Effects of Dietary Interventions on Gut Microbiota—An Up-to-Date Critical Review and Future Perspectives. Retrieved from https://www.mdpi.com/2072-6643/15/23/5005
Exploring the role of gut microbiome in male reproduction - Wang - 2022 - Andrology - Wiley Online Library. Retrieved from https://onlinelibrary.wiley.com/doi/10.1111/andr.13143
Unveiling the role of functional foods with emphasis on prebiotics and probiotics in human health: A review - ScienceDirect. Retrieved from https://www.sciencedirect.com/science/article/pii/S1756464624003396
Impact of Probiotics and Prebiotics on Gut Microbiome and Hormonal Regulation. Retrieved from https://www.mdpi.com/2624-5647/6/4/56
The gut brain axis: What it is and how to improve it. Retrieved from https://www.medicalnewstoday.com/articles/gut-brain-connection
The gut microbiota is a major regulator of androgen metabolism in intestinal contents | American Journal of Physiology-Endocrinology and Metabolism | American Physiological Society. Retrieved from https://journals.physiology.org/doi/full/10.1152/ajpendo.00338.2019
More Than a Gut Feeling: How Your Microbiome Affects Your Mood | Cognitive Enhancement. Retrieved from https://longevity.stanford.edu/lifestyle/2024/04/08/more-than-a-gut-feeling-how-your-microbiome-affects-your-mood/
Turns out your ‘gut feelings’ are real. How gut and mental health are connected | News. Retrieved from https://news.llu.edu/health-wellness/turns-out-your-gut-feelings-are-real-how-gut-and-mental-health-are-connected
Frontiers | The role of probiotics and prebiotics in modulating of the gut-brain axis. Retrieved from https://www.frontiersin.org/journals/nutrition/articles/10.3389/fnut.2023.1173660/full
The Microbiota-Gut-Brain Axis | Physiological Reviews | American Physiological Society. Retrieved from https://journals.physiology.org/doi/full/10.1152/physrev.00018.2018
Neurogastroenterology & the Gut-Brain Axis | The Institute for Functional Medicine. Retrieved from https://www.ifm.org/news/neurogastroenterology-gut-brain-axis
Microbiota–gut–brain axis and its therapeutic applications in neurodegenerative diseases | Signal Transduction and Targeted Therapy. Retrieved from https://www.nature.com/articles/s41392-024-01743-1